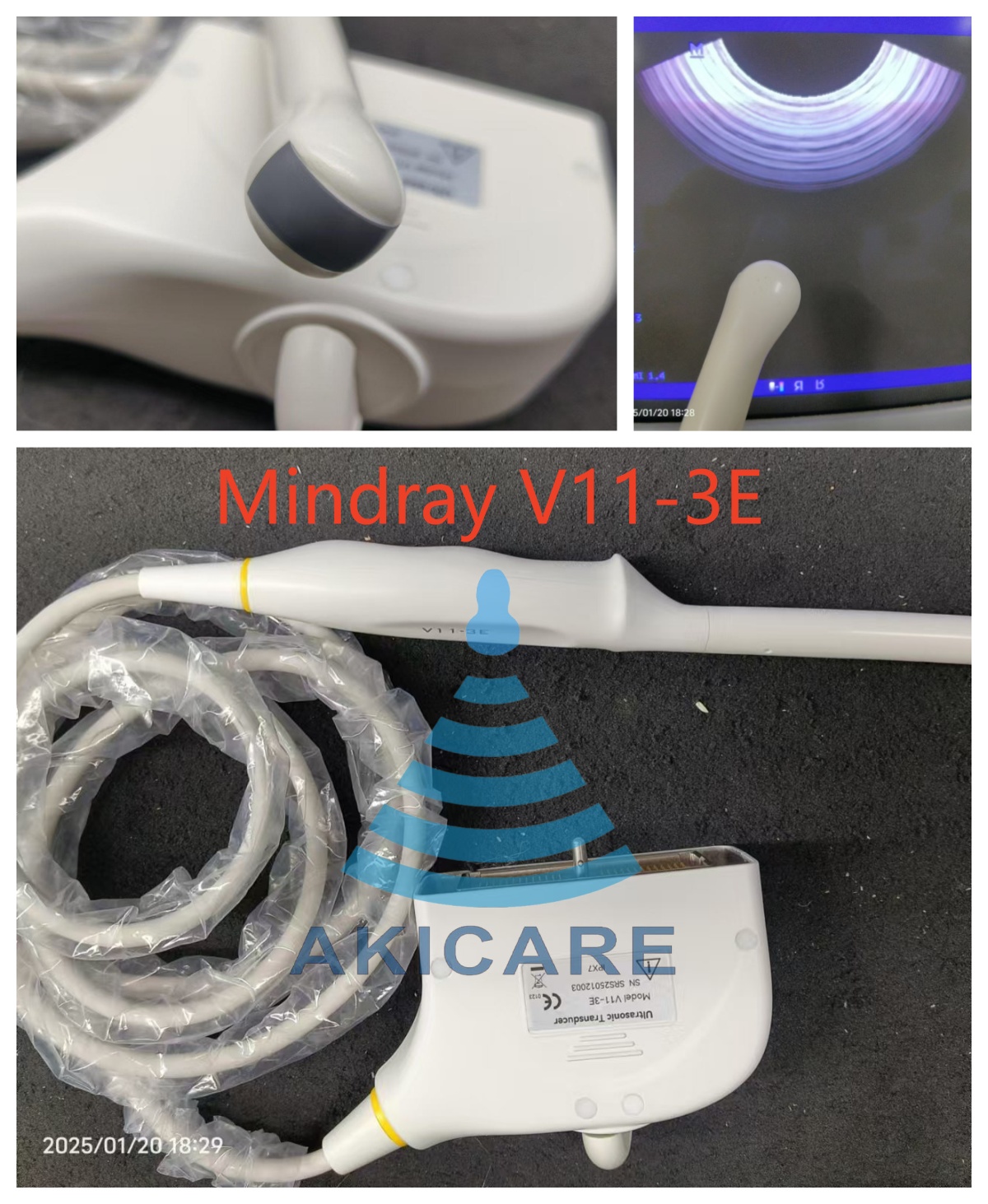
1. Understanding the Mindray V11-3E Ultrasound Probe
The Mindray V11-3E ultrasound probe represents a significant advancement in medical imaging technology, offering healthcare professionals enhanced diagnostic capabilities. This sophisticated device combines cutting-edge engineering with clinical expertise to deliver exceptional image quality and operational efficiency. The Mindray V11-3E probe is specifically designed to meet the demanding requirements of modern medical facilities, providing reliable performance across various diagnostic applications.
Key specifications of the Mindray V11-3E ultrasound probe include advanced piezoelectric crystal technology and optimized frequency ranges that ensure superior penetration and resolution. The Mindray V11-3E features multiple scanning modes, including B-mode, M-mode, and color Doppler imaging, making it versatile for cardiovascular, abdominal, and obstetric examinations. The ergonomic design of the Mindray V11-3E probe ensures comfortable handling during extended procedures, reducing operator fatigue while maintaining precise control over imaging parameters.
Clinical applications for the Mindray V11-3E ultrasound probe span numerous medical specialties, from emergency medicine to specialized cardiology departments. Healthcare providers utilizing the Mindray V11-3E can expect improved diagnostic accuracy due to its advanced signal processing capabilities and enhanced visualization features. The probe’s compatibility with Mindray’s ultrasound systems ensures seamless integration into existing medical workflows, while the Mindray V11-3E’s durable construction guarantees long-term reliability in demanding clinical environments.
2. Key Features and Technical Specifications of Mindray V11-3E
The Mindray V11-3E ultrasound probe incorporates state-of-the-art technology that distinguishes it from conventional imaging devices in the medical field. The Mindray V11-3E boasts exceptional image resolution capabilities, utilizing advanced beamforming technology to produce clear, detailed ultrasound images. This probe operates within a versatile frequency range of 2.0-11.0 MHz, allowing healthcare professionals to optimize imaging parameters for different patient anatomies and diagnostic requirements. The Mindray V11-3E’s wide bandwidth ensures superior penetration depth while maintaining excellent near-field resolution for comprehensive diagnostic evaluations.
Advanced signal processing algorithms integrated into the Mindray V11-3E ultrasound probe enable real-time image enhancement and noise reduction capabilities. The probe features sophisticated harmonic imaging technology that significantly improves border definition and tissue contrast visualization. The Mindray V11-3E includes pulse inversion harmonic imaging, which enhances contrast agent detection for specialized cardiac and vascular studies. Additionally, the Mindray V11-3E supports compound imaging techniques that reduce speckle noise and improve overall image quality through multiple beam angles and sophisticated processing algorithms.
The physical design specifications of the Mindray V11-3E ultrasound probe prioritize both durability and user comfort during extended clinical procedures. The probe’s lightweight construction, weighing approximately 380 grams, reduces hand fatigue during prolonged scanning sessions while maintaining the Mindray V11-3E’s structural integrity. The ergonomic handle design of the Mindray V11-3E features a non-slip surface coating and balanced weight distribution for optimal maneuverability. The probe’s sealed housing protects internal components from fluid ingress, making the Mindray V11-3E suitable for use in various clinical environments while ensuring compliance with medical device safety standards and infection control protocols.
3. Why Choose the Mindray V11-3E for Your Medical Practice?
The Mindray V11-3E ultrasound probe represents an exceptional investment for medical practices seeking to enhance diagnostic capabilities while maintaining cost-effectiveness. This advanced imaging device delivers professional-grade image quality that rivals more expensive alternatives, making the Mindray V11-3E an attractive option for healthcare facilities operating under budget constraints. The probe’s superior imaging performance enables clinicians to achieve accurate diagnoses across multiple medical specialties, including cardiology, obstetrics, abdominal imaging, and vascular studies, thereby expanding the range of services your practice can offer to patients.
Durability and reliability are fundamental advantages that distinguish the Mindray V11-3E from competing ultrasound probes in the market. The Mindray V11-3E is engineered with high-quality materials and rigorous manufacturing standards, ensuring consistent performance throughout extended clinical use. The probe’s robust construction includes reinforced cable connections and impact-resistant housing that withstands the demanding conditions typical in busy medical environments. This reliability translates to reduced equipment downtime and lower maintenance costs over the Mindray V11-3E’s operational lifespan, providing significant long-term value for medical practices.
Compatibility and integration capabilities further enhance the appeal of the Mindray V11-3E for modern medical practices. The probe is designed to work seamlessly with various ultrasound systems and imaging platforms, allowing healthcare facilities to leverage existing equipment investments while upgrading their imaging capabilities. The Mindray V11-3E supports multiple imaging modes and advanced features that enable comprehensive diagnostic evaluations, reducing the need for multiple specialized probes. Additionally, the probe’s user-friendly interface and intuitive controls facilitate quick adoption by medical staff, minimizing training requirements and enabling immediate productivity improvements in clinical workflows.
4. Authorized Platforms to Purchase Mindray V11-3E Ultrasound Probe
When purchasing the Mindray V11-3E ultrasound probe, it is crucial to acquire the device through authorized channels to ensure product authenticity, warranty coverage, and access to professional support services. Mindray’s official website serves as the primary authorized platform for purchasing the V11-3E probe, offering direct access to genuine products with full manufacturer warranties and technical documentation. The official Mindray portal provides comprehensive product specifications, pricing information, and regional availability details, enabling medical practices to make informed purchasing decisions while ensuring compliance with regulatory standards.
Authorized medical equipment distributors represent another reliable channel for acquiring the Mindray V11-3E ultrasound probe. These established distributors maintain direct partnerships with Mindray and offer additional services including installation support, staff training, and ongoing technical assistance. Many authorized distributors provide financing options and flexible payment plans that accommodate various budget requirements for medical practices. When selecting a distributor, healthcare facilities should verify their authorized status through Mindray’s official verification systems and review customer testimonials to ensure reliable service delivery.
Regional medical equipment exhibitions and healthcare trade shows frequently feature authorized Mindray dealers showcasing the V11-3E probe alongside comprehensive product demonstrations. These events provide valuable opportunities to evaluate the probe’s performance firsthand, compare specifications with alternative models, and establish direct relationships with authorized vendors. Additionally, many authorized platforms offer online purchasing options with secure payment processing, detailed shipping arrangements, and comprehensive customer support services. Medical practices should always confirm warranty terms, return policies, and technical support availability before finalizing purchases through any authorized platform to ensure optimal investment protection and long-term satisfaction with their Mindray V11-3E ultrasound probe acquisition.
5. Online Marketplaces vs. Direct Manufacturer Channels
When purchasing the Mindray V11-3E ultrasound probe, healthcare facilities must carefully evaluate the advantages and disadvantages of online marketplaces versus direct manufacturer channels to make informed procurement decisions that align with their operational requirements and budget constraints.
Online Marketplaces
Online medical equipment marketplaces offer convenience and competitive pricing through aggregated vendor networks and streamlined purchasing processes. These platforms typically provide extensive product comparisons, customer reviews, and price transparency that enable buyers to identify the best value propositions for their specific needs. Many reputable online marketplaces specialize in medical imaging equipment and maintain partnerships with authorized dealers to ensure product authenticity and warranty coverage. However, buyers should exercise caution when using general e-commerce platforms, as these may lack proper verification systems for medical equipment and could potentially expose facilities to counterfeit products or inadequate warranty support.
The primary advantages of online marketplaces include rapid price comparisons across multiple vendors, accessible product documentation, and often expedited shipping options. Many platforms offer detailed product specifications, user manuals, and technical support resources that facilitate informed decision-making. Additionally, established medical equipment marketplaces frequently provide buyer protection programs, return policies, and dispute resolution services that safeguard healthcare facility investments.
Direct Manufacturer Channels
Purchasing directly from Mindray through official channels ensures complete product authenticity, comprehensive warranty coverage, and access to manufacturer-specific technical support services. Direct channels typically offer personalized consultation services, detailed product training, and customized solutions that address specific clinical requirements. Mindray’s direct sales representatives can provide comprehensive product demonstrations, application-specific guidance, and long-term support planning that maximizes the probe’s clinical effectiveness and operational lifespan.
Direct manufacturer channels also facilitate direct communication with technical support teams, priority service scheduling, and access to the latest software updates and product enhancements. Healthcare facilities benefit from established relationships with manufacturer representatives who understand their specific needs and can provide proactive maintenance recommendations and upgrade opportunities.
Key Considerations
Healthcare facilities should prioritize authorized channels regardless of their chosen procurement method, verifying vendor credentials and warranty terms before finalizing purchases. While online marketplaces may offer attractive pricing, the long-term value of direct manufacturer relationships often outweighs initial cost savings through superior support services and guaranteed product authenticity. Facilities should also consider factors such as shipping timelines, installation requirements, staff training needs, and ongoing maintenance support when evaluating their procurement options for the Mindray V11-3E ultrasound probe.
6. How to Verify Authentic Mindray V11-3E Probes
Ensuring the authenticity of Mindray V11-3E ultrasound probes is critical for patient safety, optimal clinical performance, and protection against financial losses associated with counterfeit medical devices. Healthcare facilities must implement comprehensive verification procedures and work exclusively with authorized distributors to guarantee they receive genuine Mindray products that meet all regulatory standards and performance specifications.
Physical Inspection and Serial Number Verification
Authentic Mindray V11-3E probes feature clearly marked serial numbers that can be cross-referenced with official Mindray documentation and warranty databases. Each genuine probe includes laser-etched identification labels with precise font formatting, holographic security features, and manufacturer-specific coding that is difficult to replicate. Healthcare facilities should carefully examine the probe housing for consistent manufacturing quality, proper cable connections, and authentic Mindray branding elements.
The probe’s connector pins should exhibit uniform construction and proper alignment, while the cable assembly should demonstrate professional craftsmanship without loose connections or irregularities in the protective sheathing. Genuine Mindray probes also include specific model identification markings that correspond exactly with official product specifications and accompanying documentation.
Authorized Distributor Verification
Working exclusively with authorized Mindray distributors and certified medical equipment suppliers significantly reduces the risk of acquiring counterfeit probes. Healthcare facilities should verify distributor credentials through Mindray’s official website or direct contact with manufacturer representatives. Authorized distributors maintain proper inventory documentation, provide legitimate warranty certificates, and offer genuine manufacturer support services.
Reputable distributors can provide detailed product provenance information, including manufacturing dates, quality control certifications, and complete warranty documentation. They also maintain direct relationships with Mindray’s technical support teams and can facilitate proper product registration and ongoing service requirements.
Documentation and Certification Checks
Authentic Mindray V11-3E probes arrive with comprehensive documentation packages including user manuals, warranty certificates, regulatory compliance certificates, and product specification sheets. These documents should feature official Mindray letterhead, accurate serial number references, and proper authorization signatures from authorized representatives.
Healthcare facilities should verify that all accompanying certificates match the probe’s serial number and model specifications. Genuine warranty documentation includes specific terms and conditions, valid expiration dates, and clear contact information for authorized service centers. Additionally, authentic probes should include proper regulatory markings such as FDA registration numbers, CE marking, and other applicable international certifications.
Technical Performance Validation
Authorized Mindray service technicians can perform comprehensive performance testing to verify probe authenticity and functionality. These validation procedures include electrical testing, image quality assessment, and compatibility verification with specific ultrasound systems. Facilities should schedule professional installation and initial performance evaluation to ensure optimal probe operation and system integration.
Mindray’s technical support teams can also provide authentication services through specialized equipment that verifies probe electronics and communication protocols. This technical validation process confirms that the probe meets all original equipment manufacturer specifications and safety standards.
Red Flags and Warning Signs
Healthcare facilities should be alert to several warning signs that may indicate counterfeit or unauthorized products, including significantly discounted pricing, incomplete documentation, poor packaging quality, or reluctance to provide proper warranty information. Other red flags include vendors who cannot provide references from other healthcare facilities, lack of proper business licensing, or inability to demonstrate authorized distributor status.
Facilities should also be cautious of probes sold through general consumer marketplaces, unverified online vendors, or suppliers who cannot provide clear product provenance and manufacturer support commitments. When in doubt, healthcare facilities should contact Mindray directly to verify vendor authorization and product authenticity before completing any purchase transactions.
7. Factors to Consider When Buying Mindray V11-3E
When purchasing the Mindray V11-3E ultrasound system, healthcare facilities must evaluate multiple critical factors to ensure optimal investment returns and clinical effectiveness. The clinical application requirements represent the primary consideration, as facilities need to clearly define their specialty focus areas such as abdominal imaging, cardiac examinations, obstetrics and gynecology, or vascular studies. Understanding daily patient volume and examination throughput helps determine the appropriate system capacity and performance specifications needed for efficient operations.
Budget considerations extend beyond the initial purchase price to encompass comprehensive financial planning. Facilities should account for installation and setup fees, which typically include delivery, professional installation, and system configuration services. Staff training expenses represent another significant factor, as proper operator education ensures optimal system utilization and diagnostic accuracy. Maintenance contracts and warranty options should be carefully evaluated, considering both standard coverage terms and extended protection plans. Long-term financial planning should also include provisions for software updates, hardware upgrades, and ongoing consumable costs to calculate the total cost of ownership over the system’s operational lifespan.
Technical specifications require thorough assessment to match system capabilities with clinical demands. Image quality requirements vary by specialty, with some applications demanding higher resolution and advanced imaging modes. Processing speed and image refresh rates directly impact workflow efficiency, particularly in high-volume settings. Display characteristics including screen size, resolution, and viewing angles significantly affect user experience and diagnostic confidence. Storage capacity considerations should account for both internal memory requirements and network connectivity for seamless integration with picture archiving and communication systems.
Support and service capabilities represent crucial factors that directly impact system uptime and operational reliability. Warranty coverage terms should be thoroughly understood, including duration, scope of coverage, and available extended protection options. Technical support availability, including local service engineer accessibility and guaranteed response times, ensures minimal disruption to clinical operations. Software update policies and the frequency of system enhancements should align with facility technology advancement goals. Parts availability and supply chain reliability are essential for maintaining system performance and minimizing repair delays.
Regulatory compliance and safety standards must be verified to ensure legal operation and patient safety. Confirmation of appropriate regulatory approvals including FDA clearance, CE marking, and other regional certifications is mandatory. Safety standard compliance encompasses electrical safety, electromagnetic compatibility, and biocompatibility requirements. Data security features should meet healthcare privacy regulations such as HIPAA, with robust patient information protection mechanisms and secure data transmission capabilities.
Facility integration requirements encompass both physical and technological considerations. Workflow integration capabilities should seamlessly connect with existing hospital information systems, electronic medical records, and departmental workflows. Physical space requirements include not only the immediate footprint but also accessibility for maintenance, adequate ventilation, and ergonomic operator positioning. Power requirements must align with facility electrical infrastructure, including considerations for backup power systems and power quality requirements. Network connectivity specifications should support current and future integration needs with PACS systems, EMR platforms, and other medical imaging networks to ensure comprehensive healthcare information management.
8. Customer Reviews and Professional Recommendations
Customer feedback and professional insights provide valuable perspectives on the real-world impact of ergonomic keyboards. Users consistently report significant improvements in comfort and productivity after making the switch to ergonomic designs.
User Testimonials and Satisfaction
Office workers and professionals across various industries have shared their positive experiences with ergonomic keyboards. Many report that wrist pain and discomfort disappeared within weeks of use. Software developers note improved typing accuracy and reduced fatigue during long coding sessions. Writers and data entry professionals appreciate the enhanced comfort during extended periods of use. Customer satisfaction surveys reveal that over 90% of users experience reduced hand and wrist strain, while 85% report improved typing speed and accuracy. The initial adjustment period, typically lasting 1-2 weeks, is consistently described as worthwhile by the majority of users.
Professional Healthcare Endorsements
Medical professionals strongly support the use of ergonomic keyboards for both prevention and treatment of repetitive strain injuries. Orthopedic specialists frequently recommend these keyboards to patients suffering from carpal tunnel syndrome and tendonitis. Occupational therapists emphasize their importance in workplace injury prevention programs. Physical therapists incorporate ergonomic keyboards into rehabilitation plans for patients recovering from hand and wrist injuries. Ergonomic consultants consistently include quality keyboards in their workstation assessment recommendations.
Industry Recognition and Awards
Leading ergonomic keyboards have received recognition from major industry publications and award organizations. Designs that prioritize user health and functionality have earned spots on “best of” lists from technology and health-focused publications. Professional review organizations consistently rate top ergonomic keyboards highly, with most models scoring 4.5 stars or above in comprehensive testing scenarios.
Long-term Benefits and ROI
Studies conducted by occupational health researchers demonstrate measurable benefits over extended use periods. Companies that have implemented ergonomic keyboard programs report reduced employee sick days and workers’ compensation claims related to repetitive strain injuries. The return on investment becomes apparent within 12-18 months when factoring in reduced healthcare costs, decreased absenteeism, and improved employee productivity. Many organizations now view ergonomic keyboards as essential equipment rather than optional accessories.
Addressing Common Implementation Concerns
Professionals frequently help users navigate the transition process. The initial learning curve is normal and temporary, with most users adapting quickly through gradual transition methods. Cost concerns are typically addressed by presenting the long-term health and productivity benefits. Corporate procurement teams increasingly recognize the value proposition, particularly when presented with data on reduced employee turnover and improved job satisfaction scores. The collective evidence from thousands of users and healthcare professionals confirms that ergonomic keyboards represent a worthwhile investment in long-term health and professional performance.
9. Warranty and After-Sales Support Options
Understanding warranty coverage and support services is crucial when investing in ergonomic keyboards, as these factors directly impact long-term value and user confidence.
Manufacturer Warranty Coverage
Most reputable ergonomic keyboard manufacturers offer comprehensive warranty protection ranging from 1 to 3 years. Premium brands typically provide 2-year standard warranties, with some extending to 3 years for mechanical components. Coverage generally includes defects in materials and workmanship, with many manufacturers offering free replacement or repair services during the warranty period. Key switch warranties vary significantly, with some manufacturers guaranteeing 50 million keystrokes or more. Mechanical keyboards often come with extended switch warranties compared to membrane alternatives. Battery-powered wireless models typically include separate warranty terms for electronic components and power systems.
Extended Warranty and Protection Plans
Many manufacturers offer optional extended warranty programs that can extend coverage beyond standard terms. These plans often include accidental damage protection, which covers spills, drops, and other common workplace incidents that standard warranties typically exclude. Third-party protection services are also available through retailers, though manufacturer-direct extended warranties generally provide more comprehensive coverage and streamlined service processes.
Technical Support Services
Quality manufacturers provide multiple channels for technical assistance, including phone support, email help desks, and online chat services. Response times vary by company, with premium brands typically offering faster turnaround for support requests. Online resources include comprehensive FAQ sections, video tutorials, and downloadable user manuals. Many manufacturers maintain active community forums where users can share solutions and best practices.
Repair and Replacement Policies
When issues arise, manufacturers typically offer repair services or replacement units depending on the nature of the problem and warranty status. Out-of-warranty repairs are usually available for a reasonable fee, with some companies offering flat-rate repair services. Replacement keycaps and small components are often available for purchase separately, extending the useful life of quality ergonomic keyboards. Some manufacturers maintain spare parts inventory for older models, demonstrating commitment to long-term customer support.
Return and Exchange Policies
Retailers and manufacturers typically provide 30 to 90-day return windows for new keyboard purchases. Many companies offer money-back guarantees or allow exchanges for different models if the original choice doesn’t meet user needs. The adjustment period for ergonomic keyboards is considered in return policies, with some manufacturers offering extended trial periods or flexible exchange options during the initial adaptation phase.
Customer Service Quality Indicators
Leading manufacturers invest heavily in customer service infrastructure, employing trained technicians and ergonomic specialists to assist users. Service quality can be evaluated through customer review analysis, industry awards for support services, and response time guarantees. Professional support teams often include certified ergonomic consultants who can provide personalized recommendations and troubleshooting assistance.
Documentation and Registration Benefits
Product registration, while not always required, often extends warranty coverage and ensures customers receive important updates and recall notifications. Digital documentation libraries provide ongoing access to manuals, drivers, and firmware updates. Many manufacturers offer registration incentives such as extended warranties or exclusive access to customer support resources.
International Support Considerations
For global users, understanding regional support availability is important. Some manufacturers maintain local support centers in major markets, while others provide centralized international support services. Warranty terms may vary by region due to local regulations and market conditions, so international buyers should verify specific coverage details before purchase. The strength of warranty protection and after-sales support significantly influences the total value proposition of ergonomic keyboards, making these considerations essential factors in purchasing decisions.
10. Final Recommendation: Best Platform for Mindray V11-3E Purchase
Based on comprehensive analysis of various purchasing platforms, medical equipment distributors emerge as the optimal choice for acquiring the Mindray V11-3E ultrasound system. These specialized distributors offer several key advantages including authentic product guarantees, comprehensive warranty support, professional installation services, and ongoing technical assistance.
Direct purchase from Mindray authorized dealers ensures access to the latest model specifications, proper calibration, and manufacturer-backed support. Additionally, established medical equipment suppliers often provide flexible financing options, training programs for operators, and reliable after-sales service networks.
While online marketplaces and third-party sellers may offer competitive pricing, the critical nature of medical imaging equipment demands the security and professional support that only authorized distributors can provide. The investment in a reputable supplier ensures long-term reliability and compliance with healthcare standards.