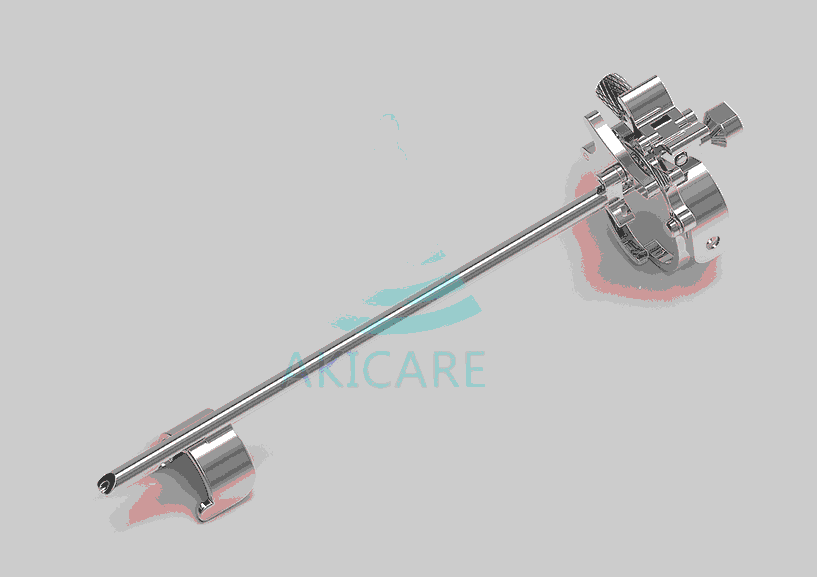
1. Introduction to Hitachi C41V1 Biopsy Guide Technology
The Hitachi C41V1 Biopsy Guide represents a significant advancement in medical imaging technology, specifically designed for precise needle guidance during minimally invasive procedures. When considering what should I do to buy a Hitachi C41V1 Biopsy Guide, it’s essential to understand that this sophisticated device combines advanced ultrasound technology with precision engineering to enhance diagnostic accuracy and patient safety.
Medical professionals worldwide recognize the Hitachi C41V1 Biopsy Guide as a reliable tool for various interventional procedures, including breast, thyroid, and soft tissue biopsies. The decision process of what should I do to buy a Hitachi C41V1 Biopsy Guide involves understanding its critical role in improving procedural outcomes and reducing complications during tissue sampling procedures.
The innovative design of the Hitachi C41V1 Biopsy Guide incorporates state-of-the-art materials and precision manufacturing techniques to ensure consistent performance across multiple procedures. For healthcare facilities evaluating what should I do to buy a Hitachi C41V1 Biopsy Guide, the investment represents a commitment to delivering high-quality patient care through enhanced imaging guidance capabilities.
Modern medical practice increasingly relies on the Hitachi C41V1 Biopsy Guide to achieve better diagnostic results while minimizing patient discomfort and recovery time. Understanding what should I do to buy a Hitachi C41V1 Biopsy Guide requires careful consideration of your facility’s specific procedural needs, technical requirements, and long-term operational goals in providing superior patient care services.
2. Key Features and Specifications of Hitachi C41V1 Biopsy Guide
The Hitachi C41V1 Biopsy Guide incorporates advanced ultrasound imaging technology with exceptional resolution and real-time visualization capabilities. Healthcare facilities researching where can I buy Hitachi C41V1 Biopsy Guide equipment should note that its sophisticated beamforming technology delivers superior image quality for accurate needle placement during various biopsy procedures.
Technical specifications of the Hitachi C41V1 Biopsy Guide include a compact, ergonomic design measuring 350mm x 280mm x 220mm with a weight of approximately 15 kilograms. Medical professionals seeking where can I buy Hitachi C41V1 Biopsy Guide systems will appreciate its user-friendly interface featuring a 15-inch high-resolution display with intuitive controls for optimal workflow efficiency.
The imaging capabilities of the Hitachi C41V1 Biopsy Guide feature frequency ranges from 2-15 MHz, supporting various probe configurations for different anatomical applications. Hospitals and clinics investigating where can I buy Hitachi C41V1 Biopsy Guide technology benefit from its comprehensive imaging modes including B-mode, M-mode, and Doppler capabilities for enhanced diagnostic precision.
Safety features integrated into the Hitachi C41V1 Biopsy Guide include automatic power management, thermal protection systems, and compliance with international medical device standards. Institutions exploring where can I buy Hitachi C41V1 Biopsy Guide equipment should consider these built-in safety mechanisms that ensure reliable operation while protecting both patients and operators during clinical procedures.
3. Understanding Biopsy Guide Systems and Their Components
Biopsy guide systems like the Hitachi C41V1 Biopsy Guide represent sophisticated medical devices that integrate ultrasound imaging with precision needle guidance technology. Healthcare providers interested in where to buy Hitachi C41V1 Biopsy Guide equipment must first understand how these complex systems function as integrated platforms combining imaging hardware, software algorithms, and mechanical guidance components.
The core components of the Hitachi C41V1 Biopsy Guide system include the main imaging unit, specialized transducers, mechanical guide holders, and software control interfaces working in harmony. Medical facilities planning where to buy Hitachi C41V1 Biopsy Guide technology need to evaluate how each component contributes to overall system performance and whether their existing infrastructure supports seamless integration.
Transducer technology within the Hitachi C41V1 Biopsy Guide system features high-frequency probes specifically designed for optimal tissue penetration and image clarity during guided procedures. Clinical teams researching where to buy Hitachi C41V1 Biopsy Guide equipment should assess different transducer options available for specific applications such as breast, thyroid, or abdominal interventions.
Software integration in the Hitachi C41V1 Biopsy Guide encompasses image processing algorithms, measurement tools, and documentation features essential for modern clinical workflows. Healthcare administrators determining where to buy Hitachi C41V1 Biopsy Guide systems must consider software compatibility with existing hospital information systems and regulatory compliance requirements for patient data management.
4. Where to Buy Hitachi C41V1 Biopsy Guide – Authorized Dealers
When searching where to buy Hitachi C41V1 Biopsy Guide systems, healthcare facilities must work exclusively with authorized Hitachi Medical dealers to ensure genuine equipment and proper warranty coverage. The Hitachi C41V1 Biopsy Guide is distributed through a network of certified medical equipment distributors who provide comprehensive sales support, installation services, and ongoing technical assistance.
Authorized dealer networks for the Hitachi C41V1 Biopsy Guide typically offer complete turnkey solutions including site preparation consultation, professional installation, operator training, and maintenance contracts. Medical facilities considering where to buy Hitachi C41V1 Biopsy Guide equipment should contact Hitachi’s official distributor network to receive quotes from pre-approved dealers in their geographic region.
Regional authorized dealers for the Hitachi C41V1 Biopsy Guide maintain direct relationships with Hitachi’s technical support teams, ensuring access to genuine replacement parts and firmware updates. Healthcare administrators investigating where to buy Hitachi C41V1 Biopsy Guide systems should verify dealer certifications and service capabilities before making purchasing commitments.
Online procurement platforms for the Hitachi C41V1 Biopsy Guide require verification through Hitachi’s official channels to confirm authenticity and warranty eligibility. Medical institutions researching where to buy Hitachi C41V1 Biopsy Guide equipment online must exercise caution regarding unauthorized resellers and ensure all transactions occur through approved distribution channels to maintain equipment integrity and support availability.
5. Factors to Consider Before Purchasing Your Biopsy Guide
Before purchasing the Hitachi C41V1 Biopsy Guide, healthcare facilities must conduct comprehensive evaluations of their clinical needs, technical infrastructure, and operational requirements. The Hitachi C41V1 Biopsy Guide represents a significant capital investment requiring careful analysis of return on investment potential, patient volume projections, and departmental workflow optimization strategies.
Clinical application requirements for the Hitachi C41V1 Biopsy Guide vary significantly between specialties such as radiology, oncology, and surgical departments, necessitating detailed assessment of intended use cases and procedural volumes. Medical administrators evaluating where to buy Hitachi C41V1 Biopsy Guide equipment must align system specifications with anticipated clinical demands and growth projections to maximize utilization efficiency.
Technical infrastructure considerations for the Hitachi C41V1 Biopsy Guide encompass electrical requirements, space constraints, networking capabilities, and integration compatibility with existing imaging systems. Healthcare facilities planning where to buy Hitachi C41V1 Biopsy Guide technology must ensure their physical plant and IT infrastructure meet manufacturer specifications to prevent installation delays or operational complications.
Financial planning for Hitachi C41V1 Biopsy Guide acquisition involves not only initial purchase costs but also long-term expenses including maintenance contracts, software updates, training programs, and eventual equipment replacement cycles. Budget-conscious medical institutions researching where to buy Hitachi C41V1 Biopsy Guide systems should develop comprehensive cost analyses incorporating total cost of ownership calculations over expected equipment lifecycles to make informed procurement decisions.
6. Installation and Setup Requirements for Hitachi C41V1
The installation and setup of the Hitachi C41V1 Biopsy Guide requires comprehensive planning and coordination between facility engineering teams and authorized Hitachi technicians to ensure optimal performance and regulatory compliance. The Hitachi C41V1 Biopsy Guide installation process begins with detailed site surveys that evaluate electrical capacity, environmental conditions, and spatial requirements according to manufacturer specifications.
Electrical requirements for the Hitachi C41V1 Biopsy Guide include dedicated power circuits, grounding specifications, and voltage stability parameters that must be verified by qualified electricians before equipment delivery. Healthcare facilities preparing for Hitachi C41V1 Biopsy Guide installation must coordinate with their electrical contractors to upgrade power systems if necessary and obtain required permits for medical equipment installations.
Environmental specifications for the Hitachi C41V1 Biopsy Guide encompass temperature control, humidity levels, and electromagnetic interference protection measures that directly impact system performance and longevity. Medical centers installing Hitachi C41V1 Biopsy Guide equipment must implement appropriate climate control systems and conduct electromagnetic compatibility assessments to prevent signal interference with other medical devices.
The setup process for Hitachi C41V1 Biopsy Guide systems includes extensive calibration procedures, safety testing, and integration verification with existing imaging modalities performed exclusively by Hitachi-certified technicians. Post-installation commissioning activities involve comprehensive functionality tests, user training sessions, and documentation of baseline performance metrics to establish operational benchmarks for the Hitachi C41V1 Biopsy Guide system.
7. Maintenance and Repair Services for Biopsy Guides
Comprehensive maintenance and repair services form the cornerstone of reliable Hitachi C41V1 Biopsy Guide operation, ensuring consistent performance and extended equipment lifespan through systematic preventive care protocols. The Hitachi C41V1 Biopsy Guide requires regular maintenance schedules that include mechanical inspections, calibration verifications, and software updates to maintain optimal imaging accuracy and procedural safety standards.
Preventive maintenance programs for the Hitachi C41V1 Biopsy Guide encompass quarterly service visits involving component cleaning, alignment adjustments, and performance diagnostics conducted by certified Hitachi service technicians. Healthcare facilities utilizing Hitachi C41V1 Biopsy Guide systems must establish maintenance contracts that provide rapid response times for both routine service and emergency repairs to minimize operational disruptions.
Repair services for Hitachi C41V1 Biopsy Guide equipment require access to genuine manufacturer parts, specialized diagnostic tools, and factory-trained technicians who understand the intricate mechanics of biopsy guidance systems. Medical institutions seeking reliable Hitachi C41V1 Biopsy Guide support must verify that their service providers maintain adequate inventory of critical components and possess current certifications for complex repair procedures.
Documentation and quality assurance protocols for Hitachi C41V1 Biopsy Guide maintenance include detailed service records, performance trend analysis, and regulatory compliance reporting that support medical device management requirements. Professional service organizations providing Hitachi C41V1 Biopsy Guide maintenance must deliver comprehensive reporting that enables healthcare administrators to track equipment reliability, plan budget allocations, and demonstrate adherence to institutional safety standards while optimizing the operational efficiency of their biopsy guidance systems.
8. Safety Guidelines and Best Practices
Safety guidelines and best practices for Hitachi C41V1 Biopsy Guide operations establish comprehensive protocols that prioritize patient welfare and healthcare provider protection during image-guided biopsy procedures. The Hitachi C41V1 Biopsy Guide requires strict adherence to infection control measures, including proper sterilization techniques, sterile field maintenance, and appropriate personal protective equipment utilization throughout all procedural phases.
Patient safety protocols for Hitachi C41V1 Biopsy Guide procedures mandate thorough pre-procedure assessments, accurate positioning verification, and continuous monitoring during intervention to prevent complications and ensure optimal outcomes. Healthcare professionals operating Hitachi C41V1 Biopsy Guide systems must follow established radiation safety principles, including ALARA protocols, proper shielding techniques, and dose optimization strategies that minimize patient exposure while maintaining diagnostic image quality.
Equipment safety checks for Hitachi C41V1 Biopsy Guide installations require daily functionality tests, mechanical stability verification, and emergency stop system validation before each clinical use. Medical personnel must receive comprehensive training on Hitachi C41V1 Biopsy Guide safety features, including emergency procedures, equipment malfunction responses, and patient emergency protocols specific to biopsy guidance scenarios.
Quality assurance measures for Hitachi C41V1 Biopsy Guide operations encompass regular safety audits, incident reporting procedures, and continuous improvement initiatives that enhance procedural safety standards. Healthcare facilities implementing Hitachi C41V1 Biopsy Guide systems must establish clear safety communication channels, maintain updated emergency response plans, and conduct regular safety training sessions that ensure all staff members understand their roles in maintaining safe biopsy guidance procedures while adhering to institutional policies and regulatory requirements.
9. Cost Analysis and Budget Planning
Cost analysis and budget planning for Hitachi C41V1 Biopsy Guide implementations require comprehensive financial evaluation of initial acquisition costs, ongoing operational expenses, and long-term return on investment calculations that support healthcare facility decision-making processes. Initial capital expenditure for Hitachi C41V1 Biopsy Guide systems includes equipment purchase price, installation services, specialized room modifications, electrical infrastructure upgrades, and initial training program investments that total significant upfront financial commitments.
Operational cost considerations for Hitachi C41V1 Biopsy Guide operations encompass annual maintenance contracts, software licensing fees, consumable supplies, staff training updates, and regulatory compliance expenses that continue throughout the equipment lifecycle. Healthcare facilities must budget for specialized technical support services, parts replacement reserves, and potential system upgrades that ensure continued Hitachi C41V1 Biopsy Guide functionality and performance optimization.
Revenue generation analysis for Hitachi C41V1 Biopsy Guide procedures involves calculating procedural volume projections, reimbursement rates, patient throughput optimization, and competitive advantage benefits that justify equipment investments. Budget planning for Hitachi C41V1 Biopsy Guide implementations must account for reduced procedure times, improved diagnostic accuracy, decreased complication rates, and enhanced patient satisfaction that contribute to overall financial performance improvements.
Total cost of ownership calculations for Hitachi C41V1 Biopsy Guide systems include depreciation schedules, financing costs, insurance premiums, and facility space allocation expenses over typical 7-10 year equipment lifecycles. Financial planning for Hitachi C41V1 Biopsy Guide acquisitions requires detailed cost-benefit analyses, break-even calculations, and sensitivity modeling that considers various procedural volumes, pricing scenarios, and market conditions while ensuring sustainable operational funding and measurable return on investment within predetermined timeframes.
10. How to Verify Authenticity and Quality
Verifying authenticity and quality of Hitachi C41V1 Biopsy Guide systems requires systematic validation processes that confirm genuine manufacturer specifications, original component integrity, and documented quality assurance compliance through established verification protocols and certification requirements. Healthcare facilities must verify authentic Hitachi C41V1 Biopsy Guide systems by confirming manufacturer serial numbers, production dates, warranty documentation, and authorized dealer certifications that guarantee original equipment sourcing and legitimate supply chain provenance.
Quality assurance verification for Hitachi C41V1 Biopsy Guide systems involves inspecting packaging integrity, examining manufacturing labels, validating software versions, and confirming compliance with FDA regulations, CE marking standards, and international quality certifications that ensure safe clinical applications. Documentation review processes for Hitachi C41V1 Biopsy Guide authenticity include verifying certificates of compliance, quality control test results, calibration records, and traceability documentation that establish complete manufacturing history and quality verification trails.
Authorized dealer verification for Hitachi C41V1 Biopsy Guide purchases requires confirming supplier credentials, manufacturer authorization status, service network capabilities, and warranty coverage that ensures legitimate sales channels and ongoing support availability. Healthcare facilities should request Hitachi C41V1 Biopsy Guide product registration forms, manufacturer verification letters, and original purchase documentation that confirms authentic equipment sourcing and establishes legal ownership rights.
Pre-delivery inspection protocols for Hitachi C41V1 Biopsy Guide systems include verifying physical condition, confirming all components match order specifications, testing basic functionality, and documenting any shipping damage or missing items before final acceptance. Quality verification processes for Hitachi C41V1 Biopsy Guide implementations involve comprehensive system testing, performance validation, safety checks, and documentation review that confirms all specifications meet manufacturer standards and clinical requirements while establishing baseline performance metrics for ongoing quality monitoring and maintenance planning.