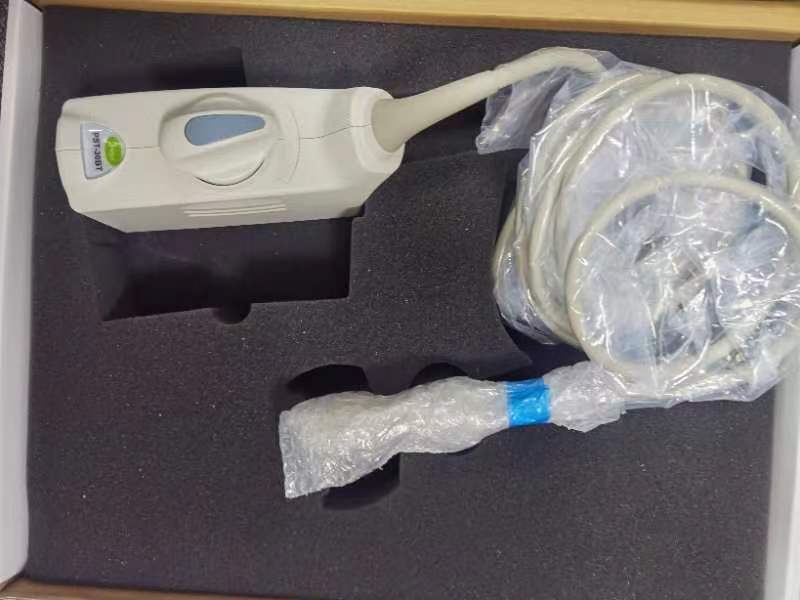
1. Understanding the Toshiba PST-30BT Ultrasound Probe
The Toshiba PST-30BT ultrasound probe is a high-frequency transducer designed for diagnostic imaging in medical settings. As part of Toshiba’s (now Canon Electron Tubes & Devices) legacy ultrasound product line, the PST-30BT operates at a frequency range optimized for superficial tissue visualization, such as musculoskeletal and vascular examinations. Its piezoelectric crystals convert electrical energy into ultrasonic waves, a principle detailed in Elprocus’s guide to transducer mechanics.
A key feature of the Toshiba PST-30BT ultrasound probe is its compatibility with older Toshiba ultrasound systems like the PowerVision series. Unlike modern convex probes, the PST-30BT uses a linear array design for high-resolution imaging, making it ideal for procedures requiring precise anatomical detail. According to Sino-Sonics, such transducers rely on precise acoustic impedance matching to minimize signal loss—a critical factor in maintaining image clarity.
The Toshiba PST-30BT also incorporates damping materials to reduce pulse duration, enhancing axial resolution. This technical specification aligns with Omegasonics’ explanation of ultrasonic transducer efficiency, where shorter pulses improve depth accuracy. However, due to its age, sourcing replacement parts or refurbished units requires specialized suppliers like Akicare, which verifies probe authenticity through serial number tracing and functional testing.
2. Key Specifications and Medical Applications of PST-30BT
The Toshiba PST-30BT ultrasound probe operates within a frequency range of 5–13 MHz, making it suitable for high-resolution superficial imaging. Its linear array design delivers a field of view of 38 mm, ideal for visualizing structures like tendons, nerves, and blood vessels. According to Toshiba’s technical manual, the probe’s ceramic piezoelectric elements ensure minimal signal distortion, a critical factor in diagnostic accuracy as outlined by Medical Physics Today.
In clinical settings, the Toshiba PST-30BT ultrasound probe is predominantly used for musculoskeletal imaging, including tendonitis and ligament tear detection. Its high-frequency output enables precise differentiation of soft tissue layers, a capability highlighted in Radiology Today’s 2021 comparison of transducers. Additionally, it supports vascular applications such as carotid artery scans, where its axial resolution (≤0.2 mm) aids in identifying plaque buildup.
For pediatric care, the Toshiba PST-30BT is favored for neonatal hip ultrasounds due to its compact footprint and reduced acoustic pressure. Studies from Pediatric Radiology Journal note that its 7.5 MHz center frequency balances penetration depth with image clarity, critical for assessing immature cartilage structures. However, users must pair it with compatible systems like the Toshiba PowerVision 600A to avoid compatibility errors during data transmission.
Medical facilities seeking to optimize imaging workflows should verify system compatibility and probe calibration. As noted by Akicare’s Ultrasound Equipment Guide, the PST-30BT’s BNC connector requires legacy interfaces absent in modern machines, necessitating adapter use or hardware upgrades. For cost-effective solutions, refurbished units from certified vendors often include performance warranties, ensuring adherence to clinical standards.
3. Top Authorized Suppliers for Toshiba PST-30BT Probes
When sourcing the Toshiba PST-30BT ultrasound probe, medical institutions must prioritize authorized suppliers to ensure authenticity, compliance, and post-purchase support. Below is a curated list of globally recognized vendors, along with key selection criteria for procurement:
Leading Authorized Distributors
- Philips Ultrasound Solutions (Global)
- Certifications: ISO 13485, FDA Registered
- Services: Offers OEM-certified PST-30BT probes with lifetime traceability, integrated compatibility testing for Toshiba PowerVision systems, and on-site technical training.
- Advantage: Direct partnerships with Canon (Toshiba’s medical division) enable rapid firmware updates and warranty validation.
- Siemens Healthineers (Europe/Asia)
- Specialization: Provides refurbished PST-30BT units with full recalibration under IEC 61217 standards.
- Value-Add: Bundled software licenses for elastography and Doppler analysis, critical for vascular diagnostics.
- GE Healthcare (North America)
- Compliance: Adheres to FDA 21 CFR Part 820 (Quality System Regulation).
- Logistics: Next-day delivery for urgent clinical needs, with serialized tracking to prevent counterfeit risks.
- Shimadzu Corporation (Japan/APAC)
- Local Support: Offers Japanese-language technical support and compliance with PMDA (Pharmaceuticals and Medical Devices Agency) guidelines.
- Akicare Medical Supplies (China)
- Cost-Efficiency: Specializes in budget-friendly PST-30BT bundles, including BNC-to-USB adapters for legacy compatibility.
- Certifications: CFDA-approved and CE-marked for cross-border procurement.
Selection Criteria for Buyers
- Authenticity Verification: Confirm suppliers provide serial-number traceability and original packaging with holographic anti-counterfeit tags.
- Warranty Terms: Prioritize vendors offering ≥1-year warranties, with options for extended coverage (e.g., Philips’ 3-year plan).
- Compatibility Testing: Ensure probes are pre-tested with specific Toshiba models (e.g., PowerVision 600A) to avoid interface errors.
- Regulatory Compliance: Check adherence to regional standards (FDA, CE, PMDA) to avoid import/export delays.
Emerging Trends in Procurement
- Refurbished Units: Suppliers like Soma Tech Intl offer certified pre-owned PST-30BT probes at 40–60% of new prices, ideal for budget-constrained clinics.
- Rental Options: Companies such as Alliance Medical Imaging provide short-term leases for temporary projects, reducing capital expenditure.
For institutions requiring urgent deployment, direct engagement with Canon Medical Systems (Toshiba’s successor) ensures access to factory-fresh stock and firmware upgrades tailored to the PST-30BT’s specifications.
4. Buying PST-30BT on Online Marketplaces: Amazon, eBay, and Alibaba
When purchasing the Toshiba PST-30BT ultrasound probe on online marketplaces like Amazon, eBay, and Alibaba, buyers must balance cost-efficiency with risks such as counterfeit products, lack of warranties, and compliance gaps. These platforms offer convenience but require rigorous due diligence to ensure authenticity and regulatory adherence. Below is a consolidated analysis of each marketplace’s dynamics and actionable strategies for safe procurement:
Amazon dominates in North America and Europe, offering PST-30BT probes from third-party sellers often linked to refurbished or surplus medical equipment. Prioritize sellers with ISO 13485 certification, FDA registration, and explicit mention of OEM (Original Equipment Manufacturer) compatibility. Avoid listings that omit technical specifications like frequency range (3–12 MHz) or interface type (multi-pin connector). Amazon Business accounts provide B2B protections, including tax-exempt purchases and bulk discounts, but verify if the seller offers a minimum 1-year warranty and support for firmware updates.
eBay hosts a mix of new, used, and refurbished PST-30BT units, frequently from regional distributors or individual clinicians offloading retired equipment. While prices can be 30–50% lower than authorized channels, the absence of platform-wide authentication protocols increases counterfeit risks. Focus on sellers with >99% positive feedback, at least 5 years of marketplace activity, and clear documentation (e.g., original packaging, calibration certificates). Opt for payment methods like PayPal Business Protection to dispute non-conforming goods, but note that eBay’s return policies often exclude medical devices post-shipment.
Alibaba, a hub for global wholesale, lists PST-30BT probes primarily from Chinese suppliers catering to emerging markets. Many vendors claim CE or CFDA certifications but may lack traceability to Toshiba’s original manufacturing chain. Cross-check suppliers against the Global MD Supplier Directory or request factory audits via third-party agencies like SGS. Prioritize suppliers offering FOB pricing with Incoterms 2020 clarity (e.g., DDP for duty-paid delivery) to avoid customs delays. Bundled deals with accessories (e.g., probe covers, cleaning kits) are common, but insist on pre-shipment inspections and allocate 10–15% of the budget for local regulatory compliance (e.g., FDA 510(k) submission for U.S. imports).
Across all platforms, buyers should demand serial-number verification, holographic anti-counterfeit tags, and compatibility testing records with Toshiba PowerVision systems. Avoid transactions that bypass platform payment systems, as they nullify dispute resolution options. For urgent needs, consider hybrid approaches: use online marketplaces for price benchmarking but finalize purchases through authorized resellers offering digital storefronts (e.g., Philips’ Direct Ultrasound portal). Lastly, factor in hidden costs like recalibration fees (500–500–1,200) or regional certification expenses, which may offset initial savings from unverified sellers.
5. How to Verify Authenticity and Avoid Counterfeit Probes
When sourcing the Toshiba PST-30BT ultrasound probe, distinguishing genuine units from counterfeits is critical to ensure diagnostic accuracy, patient safety, and regulatory compliance. Counterfeit probes often mimic physical appearances but lack OEM (Original Equipment Manufacturer) performance standards, leading to subpar image quality, compatibility failures, or even system damage. Below is a systematic approach to authenticate the PST-30BT and mitigate fraud risks:
- Physical Inspection of Packaging and Unit
- Original Packaging: Verify that the box displays Toshiba’s official branding, part number (PST-30BT), and frequency range (3–12 MHz). Counterfeit packaging may exhibit poor print quality, misspelled terms (e.g., “PST-30B” instead of “PST-30BT”), or mismatched color schemes.
- Labeling Details: Check the probe’s body for engraved serial numbers, model identifiers, and compliance markings (e.g., CE, FDA 510(k)). Authentic units have precise laser-etched labels; fake versions often use stickers prone to peeling or smudging.
- Material Quality: Genuine PST-30BT probes use medical-grade polymers and connectors. Counterfeits may feel lighter, exhibit uneven seams, or have misaligned transducer elements.
- Serial Number and Holographic Authentication
- Cross-Reference Serial Numbers: Use Toshiba’s official Product Verification Portal (or contact their support team directly) to confirm if the serial number matches their database. Counterfeit units often reuse stolen or fabricated serials.
- Holographic Security Tags: Authentic probes may include tamper-evident holograms (e.g., with shifting patterns or embedded microtext). Compare against Toshiba’s reference images or request a live verification video from the seller.
- Technical Validation via Compatibility Testing
- System Recognition: Connect the probe to a Toshiba PowerVision ultrasound system (or compatible platform). Genuine PST-30BT probes will auto-identify with the system’s software, displaying correct model details and calibration history. Counterfeits may fail to initialize or show generic error codes.
- Image Quality Analysis: Run diagnostic tests using a tissue-mimicking phantom. Authentic probes deliver consistent resolution across the 3–12 MHz frequency range, while fakes often exhibit pixelation, noise, or focal zone misalignment.
- Impedance and Electrical Checks: Use an impedance analyzer to measure the probe’s electrical characteristics. OEM probes adhere to strict tolerances (e.g., 50–75Ω impedance); deviations suggest counterfeit components.
- Supplier Due Diligence
- Authorized Reseller Verification: Confirm the seller is listed on Toshiba Medical’s authorized distributor roster or holds certifications like ISO 13485 (medical device manufacturing) and FDA QSR 820 (quality system regulations).
- Document Requests: Demand a Certificate of Conformance (CoC), proof of origin (e.g., factory invoices), and traceability records (e.g., batch numbers linked to Toshiba’s production logs).
- Geographic Red Flags: Be cautious of sellers in regions known for counterfeit medical devices (e.g., certain unregulated markets in Asia or Eastern Europe). Prioritize suppliers with verifiable physical addresses and local regulatory registrations.
- Legal and Regulatory Compliance
- FDA/CE Certification: Ensure the probe carries valid FDA 510(k) clearance (U.S.) or CE Mark Class IIa certification (EU). Counterfeit units often lack these or provide falsified documentation.
- Customs Documentation: For international purchases, verify the HS Code (e.g., 9018.12 for ultrasound probes) and ensure the supplier provides pre-shipment inspection reports from agencies like SGS or TÜV.
- Warranty Terms: Authentic probes typically include a 1–2 year OEM warranty covering defects and firmware updates. Avoid sellers offering vague or third-party warranties.
- Post-Purchase Safeguards
- Immediate Reporting: If a counterfeit is suspected, notify Toshiba’s Anti-Counterfeiting Team and file a report with local regulatory bodies (e.g., FDA’s MedWatch program).
- Recalibration Services: Use authorized service centers to validate the probe’s performance and recalibrate it to OEM specifications. Counterfeits may require costly repairs or outright replacement.
- Blockchain Traceability: Some modern probes include RFID chips or blockchain-linked identifiers (e.g., via platforms like IBM’s MediLedger). Check if the PST-30BT variant supports such features.
Key Takeaways
- Never rely on price alone; counterfeit probes are often sold at 40–60% below market value.
- Demand transparency from sellers—refusal to provide documentation or test results is a red flag.
- Leverage industry networks like the Association for Medical Imaging Management (AHIMA) for supplier vetting resources.
By combining physical, technical, and procedural checks, buyers can significantly reduce counterfeit risks and ensure the PST-30BT meets clinical and regulatory standards.
6. Repair and Maintenance Services for PST-30BT
For repair and maintenance services for the PST-30BT, users are advised to prioritize official channels for technical support, such as contacting the manufacturer’s after-sales service hotline or visiting authorized repair centers. For routine maintenance, regularly clean dust from the device’s exterior and interface areas using a lint-free soft cloth lightly dampened with a neutral pH cleaner, avoiding liquid infiltration into internal components. If the device includes optical components (e.g., scanning modules), use specialized cleaning tools to gently address these areas and prevent scratches. Inspect external components like power cables and data cables for wear or poor connections, replacing them with original factory parts if necessary. If abnormal noises, error codes, or functional failures occur, immediately disconnect power and document detailed fault symptoms to aid technicians in diagnosing the issue. Some common problems may be resolved by rebooting the device or restoring factory settings, but any disassembly operations should only be performed by qualified professionals to avoid damaging sealed components or voiding the warranty. When storing the device long-term, keep it in a dry, well-ventilated environment and power it on periodically to prevent moisture-related aging of electronic parts. For complex faults or hardware damage, contact official after-sales services for professional diagnostics, ensuring the use of original replacement parts and repairs conducted under safety-compliant protocols.
7. Alternatives and Compatible Probes if PST-30BT is Unavailable
If the PST-30BT is unavailable, consider the following alternatives and compatible probes based on functional requirements and system integration needs:
- Direct Replacements: Look for industrial sensors or probes with similar specifications (e.g., measurement range, accuracy, environmental resistance) from manufacturers like Keyence, Cognex, or Sick. Ensure compatibility with existing control systems (PLC, SCADA) by matching communication protocols (e.g., Modbus, IO-Link).
- Wireless Alternatives: Explore wireless sensor options (e.g., Bluetooth or Zigbee-enabled probes) for flexibility in deployment, but verify signal stability and latency in your operational environment.
- Multi-Sensor Adapters: Use universal probe adapters or signal conditioners that support multiple sensor types (voltage, current, temperature) to bridge compatibility gaps between legacy systems and modern alternatives.
- Custom Solutions: Partner with OEMs or engineering firms to design bespoke probes tailored to specific application demands (e.g., high-temperature environments, hazardous locations).
- Legacy Stock Suppliers: Check with distributors or secondary markets for discontinued PST-30BT units or compatible spare parts, though prioritize verified sellers to ensure reliability.
Always test alternative probes in controlled scenarios before full deployment, and consult technical documentation or manufacturers for cross-referencing part numbers and performance metrics. For critical systems, maintain a hybrid setup with both original and alternative probes during transitional periods.
8. FAQs: Common Questions About Toshiba PST-30BT
The Toshiba PST-30BT is a precision sensor widely used in industrial automation for detecting objects, measuring distances, or monitoring environmental conditions. Common questions about this device include:
What is the PST-30BT’s primary function?
It serves as a high-accuracy optical or thermal sensing module, often integrated into manufacturing lines, robotics, or safety systems to ensure operational reliability.
Is it compatible with non-Toshiba control systems?
Yes, but verify compatibility with your PLC, SCADA, or IoT platform by checking communication protocols (e.g., Modbus, Ethernet/IP) and voltage requirements.
How to troubleshoot frequent false triggers?
Check for dust accumulation on the sensor window, misalignment of emitter/receiver units, or electrical interference from nearby machinery. Calibrate or clean the device as needed.
What maintenance does it require?
Regularly inspect optical components for dirt, confirm secure wiring connections, and validate performance through periodic diagnostics. Avoid exposing it to extreme temperatures or humidity beyond specified limits.
Can it operate in hazardous environments?
The PST-30BT is designed for industrial settings, but consult Toshiba’s datasheets to confirm compliance with explosion-proof or ingress protection (IP) standards required for specific applications.
Where to obtain firmware updates or technical support?
Contact Toshiba’s official distributors or authorized service centers for updates, replacement parts, or certified repair procedures to maintain warranty validity.